 The group of halogens is the only periodic table group that contains elements in three of the main states of matter at standard temperature and pressure, though not far above room temperature the same becomes true of groups 1 and 15, assuming white phosphorus is taken as the standard state. When halogens react with metals, they produce a wide range of salts, including calcium fluoride, sodium chloride (common table salt), silver bromide and potassium iodide. The word "halogen" means "salt former" or "salt maker". 
In the modern IUPAC nomenclature, this group is known as group 17. 
This was impressive, because he left gaps for elements that hadn’t been discovered yet and he did this before we fully understood the structure and composition of atoms.The halogens ( / ˈ h æ l ə dʒ ə n, ˈ h eɪ-, - l oʊ-, - ˌ dʒ ɛ n/ ) are a group in the periodic table consisting of six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and the radioactive elements astatine (At) and tennessine (Ts), though some authors would exclude tennessine as its chemistry is unknown and is theoretically expected to be more like that of gallium. Several scientists arranged the elements they knew about in different ways before Dmitri Mendeleev arranged them as they are today. 
The periodic table is useful to chemists because of the way the elements are arranged but this didn’t happen quickly or by chance. They are usually relatively hard and are often used as catalysts (they help speed up chemical reactions, without being used up in the reactions themselves). The transition elements are metals and often react with other elements to form brightly coloured compounds. The block in the middle of the table, beginning in the fourth period, contains the transition elements. For example, you’ll find metals on the left-hand side and non-metals on the right. This means that, however many elements might be listed in an equation, you’ll always be able to distinguish between them.Īs you move along each row (or period), there are repeating patterns in the chemical and physical properties of the elements in each one. When you write the symbol for an element, it’s really important that the first letter is a capital and the second letter (if it has one) is lower case. Giving them symbols makes this process clearer, especially as these symbols are recognised all over the world, even if the names themselves vary in different languages. Classroom presentation as MS Powerpoint or pdfĭOWNLOAD ALL Reading and writing chemical symbolsĬhemists spend a lot of time talking and writing about elements: what they’re like, how they react, how they change.Teacher notes, printable cards and answers as MS Word or pdf. 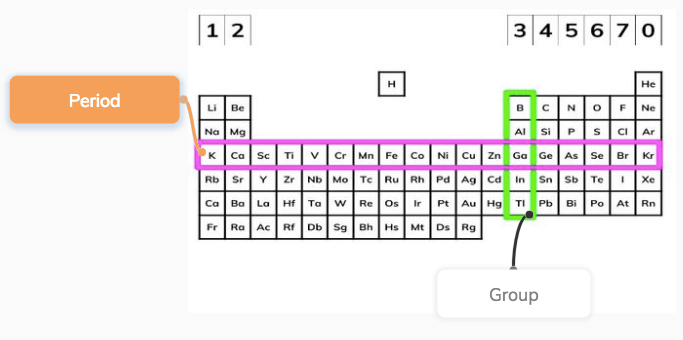
Student activity and worksheet as MS Word or pdf.Learners can use the card sort included in the teacher notes or get the information from the presentation slides to build their own miniature table with just nine elements and three groups. Walk in the footsteps of the chemists who organised the modern periodic table of elements. Use the accompanying activity to organise unknown elements using their similarities, physical properties, appearance and reactivity. Alternatively print it and use as a handout. Display the poster in your classroom or on a projector. Infographic poster and fact sheet, student worksheet, card sort and classroom presentation slides for organising elements activity. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
